The design and green synthesis of novel benzotriazoloquinolinyl spirooxindolopyrrolizidines: antimycobacterial and antiproliferative studies†
Abstract
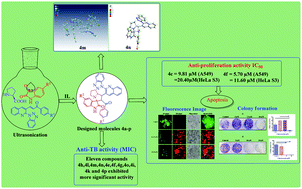
Herein, we reported the design and synthesis of a novel series of potent antimycobacterial (anti-TB) and antiproliferative benzotriazoloquinolinyl spirooxindolopyrrolizidine derivatives via an expeditious green approach achieved using an ionic liquid ([Bmim]BF4) under ultrasonication. The title compounds 4a–p were well characterized and evaluated for their in vitro anti-TB and antiproliferative activities. In anti-TB screening, eleven compounds, namely, 4h, 4l, 4m, 4n, 4e, 4f, 4g, 4o, 4i, 4k and 4p exhibited more significant anti-TB activities with the MIC values of 1.27 μM, 1.24 μM, 1.21 μM, 1.13 μM, 2.62 μM, 2.55 μM, 2.38 μM, 2.38 μM, 5.00 μM, 4.88 μM and 4.56 μM, respectively, compared to the standard drug ethambutol (MIC: 7.64 μM). With regard to antiproliferative activity, 4f exhibited more significant antiproliferative activity against the A549 (IC50: 5.7 μM) and HeLa S3 (IC50: 11.6 μM) cell lines when compared to the standard drug cisplatin. Similarly, 4c displayed equal antiproliferative activity against the A549 (IC50: 9.81 μM) and HeLa S3 (20.41 μM) cell lines. At the same time, 4c and 4f also induced typical apoptotic morphological features like cell death and inhibition of the colony formation of the A549 cells at lower concentrations. In addition, the potent anti-TB and antiproliferative compounds (4h, 4l, 4m, 4n, 4e, 4g, 4o, 4i, 4k, 4p, 4c, and 4f, respectively) displayed less toxicity against normal RAW 264.7 cells and were considered as promising for anti-TB and antiproliferative activities.


 Please wait while we load your content...
Please wait while we load your content...