Use of a bis-1,2,3-triazole gelator for the preparation of supramolecular metallogels and stabilization of gold nanoparticles†
Abstract
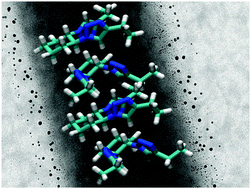
In this work we have applied a double isosteric substitution of the amide groups presented in the gelator N,N′-((1S,2S)-cyclohexane-1,2-diyl)didodecanamide (C12-Cyc) by 1,2,3-triazole rings affording the corresponding isosteric gelator (click-C12-Cyc). This approach enabled the preparation of different metallogels and fine-tuning their properties. In particular, C12-Cyc was found to form stable and reversible metallogels with PdII and AuIII in DMSO and DMF, whereas click-C12-Cyc formed metallogels in these solvents only in the presence of AuIII. Comparing the gels made of C12-Cycvs.click-C12-Cyc in the presence of HAuCl4·3H2O, the former showed higher Tgel values and 5–8 times slower gelation kinetics. Furthermore, the incorporation of Et3N as reducing agent in the formulations facilitated the in situ formation of non-aggregated gold nanoparticles embedded in the viscoelastic networks made of either pure isosteres or with an equimolar mixture of click-C12-Cyc and C12-Cyc. So-formed NPs showed similar size distribution regardless the gold concentration and the nature of the gelator system.


 Please wait while we load your content...
Please wait while we load your content...