A facile synthesis method for fabrication of LaFeO3/g-C3N4 nanocomposite as efficient visible-light-driven photocatalyst for photodegradation of RhB and 4-CP
Abstract
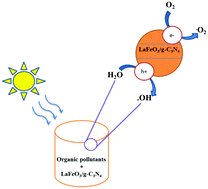
The objective of this paper was to prepare, characterize and evaluate the degradation efficiency of organic pollutants on a perovskite-type LaFeO3-coupled graphitic carbon nitride (g-C3N4) photocatalyst under visible light irradiation. The photocatalyst was synthesized by a simple calcination method. The physical and photophysical properties of the LaFeO3/g-C3N4 composite photocatalyst with various weight ratios of LaFeO3 were characterized by X-ray diffraction (XRD), transmission electron microscopy (TEM), Fourier transform infrared (FT-IR) spectroscopy, X-ray photoelectron spectroscopy (XPS), N2 adsorption–desorption isotherm measurement, UV-vis diffuse reflectance spectroscopy (DRS), photoluminescence (PL) spectroscopy, and electrochemical measurements. According to the flat band potential estimated by the Mott–Schottky plot and the optical band gap measurements, both semiconductors (LaFeO3 and g-C3N4) can absorb visible light and have band edge positions that allow the transfer of photoelectrons from g-C3N4 to LaFeO3. As compared to g-C3N4, the C 1s and N 1s peaks of the LaFeO3/g-C3N4 composite showed an obvious shift to lower binding energy, implying the existence of a synergistic effect between g-C3N4 and LaFeO3. Furthermore, photocurrent measurement and electrochemical impedance spectroscopy revealed the higher photo-induced charge carrier separation efficiency of the LaFeO3/g-C3N4 composite. The results show that the visible light activity of the composite photocatalyst LaFeO3/g-C3N4 for the degradation of Rhodamine B (RhB) and 4-chlorophenol (4-CP) is higher than that of pure LaFeO3 and g-C3N4, respectively. However, the main reason for the enhanced activity was attributed to the interfacial transfer of photogenerated electrons and holes between LaFeO3 and g-C3N4, leading to the effective charge separation in the composite, inhibited recombination of electron–hole pairs and, finally, enhanced photocatalytic performance of the composite. It was found that the holes, hydroxyl radicals (•OH) and superoxide radical ions (O2−) are the main reactive species in the degradation reaction of RhB and 4-CP over the LaFeO3/g-C3N4 composite photocatalyst. More importantly, on the basis of the estimated conduction band (CB) from the Mott–Schottky plots, LaFeO3 and the composite show no activity toward hydrogen production under visible light. This work can be applied for the production of other visible-light-responsive photocatalysts based on g-C3N4 that have potential in environmental purification applications.


 Please wait while we load your content...
Please wait while we load your content...