G-Quadruplex based hydrogels stabilized by a cationic polymer as an efficient adsorbent of picric acid†
Abstract
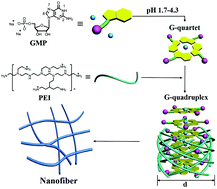
Hydrogels based on G-quadruplexes (G-hydrogels) were prepared using guanosine 5′-monophosphate disodium salt, GMP, with a hyperbranched poly(ethylenimine), PEI, containing abundant –NH2 groups. The cation–dipolar interaction between Na+ ions and GMP-quartets and the hydrogen bonding between the phosphate groups of GMP and the –NH2 groups of PEI gave rise to the hydrogels at an appropriate pH range of 1.7–4.3. X-ray diffraction (XRD) measurements and fluorescence enhancement of Thioflavin T (ThT) indicated the formation of the G-quadruplexes. The G-hydrogel showed high adsorption capacity for the contaminant, picric acid (PA), in water, ascribed to its plentiful –NH2 groups in the network structures.


 Please wait while we load your content...
Please wait while we load your content...