Synthesis and characterization of Pt(ii)-based potent anticancer agents with minimum normal cell toxicity: their bio-activity and DNA-binding properties†
Abstract
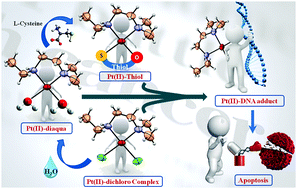
A Pt(II)-based complex [Pt(DMEEDA)Cl2], 1 (DMEEDA = N,N-dimethyl-N′-ethylethylenediamine), was synthesized, where DMEEDA acted as the (N,N) bidentate carrier ligand. The diaqua complex [Pt(DMEEDA)(H2O)2](NO3)2, 2, was obtained by the hydrolysis of dichloro complex 1. Further, its substituted complex [Pt(DMEEDA)(L-cys)](NO3), 3, and [Pt(DMEEDA)(N-ac-L-cys)], 4 (where L-cys = L-cysteine and N-ac-L-cys = N-acetyl-L-cysteine), were synthesized with amino acid L-cysteine and its acetyl derivative. The complexes were characterized via different spectroscopic techniques, such as UV-Vis, FT-IR, 1H NMR and ESI-MS. The structure of complex 1 was determined via X-ray diffraction studies to be an orthorhombic crystal system with cell parameters a = 7.9970(2) Å, b = 16.9851(7) Å, c = 16.0460(7) Å and α = 90(°); β = 90(°); γ = 90(°) and space group (pccn). The DNA-binding properties of the complexes were investigated via gel electrophoresis, and DNA-binding constants were obtained from UV-Vis spectroscopic and fluorescence titration methods. Congruent with a significant increase in DNA fragmentation, the growth inhibition potential of the synthesized Pt(II) complexes has been remarkably higher in the hepatocarcinoma cell line (HepG2), as compared to cisplatin. Moreover, these complexes show minimal toxicity to mouse primary hepatocytes and RAW 264.7 cells in vitro.


 Please wait while we load your content...
Please wait while we load your content...