Impact of the Schiff base ligand substituents on the solid state and solution properties of eleven iron(iii) complexes†
Abstract
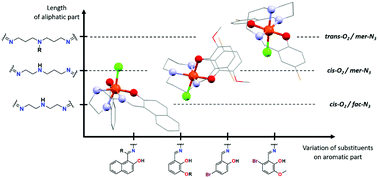
Investigations on a series of mononuclear iron(III) Schiff base complexes of the general formula [Fe(L)Cl]·S (where L2− is a Schiff base ligand anion, S is a solvent molecule) are reported. Derivatives of salicylaldehyde or 2-hydroxy-1-naphthaldehyde were used in combination with linear either symmetrical or non-symmetrical aliphatic triamines to synthesize the Schiff base ligands and subsequent reactions with iron(III) chloride afforded various [Fe(L)Cl] complexes. Compounds were characterized by conventional techniques and the crystal structures of all complexes were determined as well. A structural study revealed the relationship between the spatial arrangement of the N3O2-donor atom set and the length of the aliphatic part of the Schiff base ligand. Magnetic and EPR investigations confirmed the high spin state behaviour in all reported compounds and their analysis allowed quantification of the spin Hamiltonian parameters. Formation of hydrogen-bonded pseudodimers with an antiferromagnetic exchange coupling was found in the complexes with the shortest aliphatic part of the Schiff base ligands. Experimental exchange coupling constants were in good agreement with those obtained from DFT calculations and correlated with the strength of the hydrogen bonds. Cyclic and square wave voltammetry techniques were employed to investigate the redox properties of the reported compounds and the experimental redox potentials were compared with those obtained from the DFT calculations.


 Please wait while we load your content...
Please wait while we load your content...