Synthesis, antiviral and antibacterial activities and action mechanism of penta-1,4-dien-3-one oxime ether derivatives containing a quinoxaline moiety†
Abstract
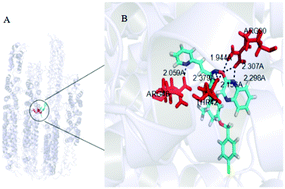
A series of penta-1,4-dien-3-one oxime ether derivatives containing a quinoxaline moiety were synthesized and their antibacterial and antiviral activities were evaluated. Bioassay activity indicated that some of the compounds displayed significant antibacterial and antiviral activities. In particular, some title compounds were found to show remarkable antiviral activities against tobacco mosaic virus (TMV). Compound 6i showed remarkable curative, protective and inactivation activity against TMV, with a 50% effective concentration (EC50) of 287.1, 157.6 and 133.0 μg mL−1, respectively. These results were better than or comparable to those of ningnanmycin (356.3, 233.7 and 121.6 μg mL−1, respectively). Microscale thermophoresis (MST) also showed that the binding of compound 6i to TMV coat protein (TMV-CP) gave a Kd value of 0.115 ± 0.092 μmol L−1, which was better than that of ningnanmycin (0.523 ± 0.254 μmol L−1). Meanwhile, the EC50 values of compound 6k against Xanthomonas axonopodis pv. Citri (Xac) and Xanthomonas oryzae pv. oryzae (Xoo) were 16.8 and 33.4 μg mL−1 respectively, and that of compound 6i against Ralstonia solanacearum (Rs) was 33.9 μg mL−1. These results were better than those of bismerthiazol (44.3, 42.5 and 62.4 μg mL−1, respectively). The mechanism of antibacterial action of compound 6k against Xac was analysed through scanning electron microscopy (SEM). This study indicated that the title compounds are valuable in the search for novel agrochemicals.


 Please wait while we load your content...
Please wait while we load your content...