Luminescent imidazolium–naphthalene salts in liquid and solid states†
Abstract
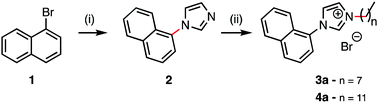
This article describes highly luminescent ionic compounds in liquid and crystalline states, where a naphthalene moiety is conjugated to an imidazolium center decorated with alkyl chains of different lengths. We have characterized, by X-ray diffraction of a single crystal and the liquid, that these compounds are organized in rod-shaped assemblies. The formation of this molecular architecture is governed by ionic interactions that dominate those of the lesser π–π and van der Waals interactions. Consequently, aromatic naphthalene rings are confined to the environment of alkyl chains (diluted in an apolar solvent), thus avoiding the extinction of luminescence by the formation of chromophore excimers, as shown in photophysical data.


 Please wait while we load your content...
Please wait while we load your content...