New anthrahydrazone derivatives and their cisplatin-like complexes: synthesis, antitumor activity and structure–activity relationship†
Abstract
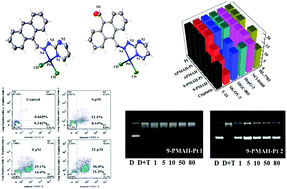
Based on the characteristic anthrahydrazone pharmacophore, two new anthrahydrazone derivatives (9-PMAH and APMAH) were synthesized, which further afforded two corresponding cisplatin-like Pt(II) complexes (9-PMAH-Pt and APMAH-Pt). Their chemical structures were characterized by IR, ESI-MS, elemental analysis and X-ray single crystal diffraction method. Their in vitro cytotoxicity towards a series of human tumor cell lines and the normal liver cell line HL-7702 were screened and shown in IC50 values and compared with that of cisplatin. 9-PMAH-Pt showed higher cytotoxicity against MGC-803 and HepG-2 than the other tested compounds including cisplatin, while it showed lower toxicity on HL-7702 than cisplatin. The structure–activity relationship from the in vitro cytotoxicity study indicated that the substitution of –CHO on the opposite C10 of anthrahydrazone was unfavorable to higher cytotoxicity, which might be due to its steric hindrance considering the planar aromatic anthracene. Further studies demonstrated that 9-PMAH-Pt induced cell apoptosis in both MGC-803 and T-24 cells by arresting the cell cycle at the G2/M phase, which was totally different for cisplatin, suggesting the different antitumor efficacies of this platinum complex and cisplatin. On the molecular level, it was suggested that 9-PMAH-Pt could bind with DNA via an intercalative mode, likely competing with GelRed in similar intercalative binding sites. 9-PMAH-Pt also effectively inhibited the activity of TOPO I, acting as a TOPO I suppressor. The results of this work suggest a new type of platinum complex bearing anthrahydrazone ligands to form a new bifunctional antitumor candidate in comparison with the classic platinum drugs.


 Please wait while we load your content...
Please wait while we load your content...