A new ratiometric, colorimetric and ‘‘turn-on’’ fluorescent chemosensor for the detection of cyanide ions based on a phenol–bisthiazolopyridine hybrid†
Abstract
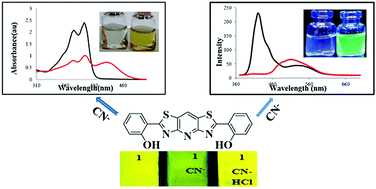
A novel C2-symmetric ratiometric fluorescence and colorimetric cyanide (CN−) sensor 1 based on a phenol–bisthiazolopyridine hybrid was synthesized and characterized. Among the various screened anions, this chromogenic receptor 1 showed only a distinct visible color change from colorless to yellow and blue to green fluorescence toward CN− in both pure methanol and aqueous methanol. The ‘‘turn-on’’ ratiometric fluorescence (emission shift = 95 nm) and colorimetric (absorbance shift = 45 nm) detection of CN− can be attributed to the deprotonation of the OH groups of 1, as evidenced by OH− and 1H NMR experiments. The binding mode of 1 with CN− was determined to be a 1 : 1 stoichiometry through a Job plot. Probe 1 was also highly sensitive (LOD = 75 nM) as measured by ratiometric fluorescence (I420/I515 nm) methods. Moreover, the reversibility of the deprotonated 1 by HCl in both solution and the solid state (TLC paper test strips) was successful. In general, probe 1 is a promising CN− indicator in terms of its ease-of-use, selectivity, sensitivity, rapid response (<1 s), reversibility and test kit application.


 Please wait while we load your content...
Please wait while we load your content...