A universal and controllable strategy of constructing transition-metal nitride heterostructures for highly enhanced bifunctional electrocatalysis†
Abstract
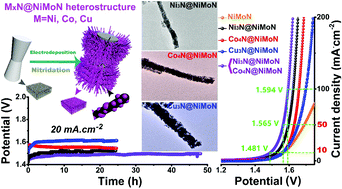
The transition-metal nitride heterostructure has attracted increasing attention due to its unique structure and amazing synergetic properties, enabling it to synchronously achieve bifunctional applications. However, the classical bifunctional electrocatalysts usually show outstanding activity for one half-reaction at the expense of the ability of another half-reaction, thus giving a moderate performance for overall water splitting. In this work, a family of transition-metal nitride heterostructures (MxN@NiMoN, M: Ni, Co and Cu) were synthesized by a general and effective strategy to address the shortcomings of classical bifunctional electrocatalysts. These materials exhibited excellent performances for both hydrogen and oxygen evolution reaction (HER/OER) that were superior or comparable to those of noble metals. Boosted electrocatalytic overall water splitting was also achieved with only a low cell voltage of 1.481 V to drive 10 mA cm−2. Moreover, the excellent durability of 2 days illustrated superior value in practical applications. This work provides fundamental insight towards designing and optimizing advanced materials for renewable energy applications.


 Please wait while we load your content...
Please wait while we load your content...