Theoretical insights into the structural, photophysical and nonlinear optical properties of phenoxazin-3-one dyes†
Abstract
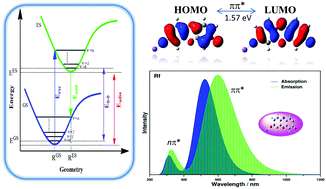
Here we investigate the structural, photophysical and nonlinear optical (NLO) properties of phenoxazin-3-one dyes, resazurin (Rz) and resorufin (Rf), by performing quantum chemical calculations using the DFT and TDDFT methods. The UV-vis absorption and fluorescence emission spectra of these dyes are computed at the BPW91/6-31+G(d,p) level in aniline solvent, and the wavelength maxima (λmax) are found to be in good agreement with the experimental results. The Stokes shift observed for these dyes is indeed overestimated by our method (TD-BPW91) compared to the experiment. The first lowest allowed and most dominant transitions for both molecules are found to be the HOMO → LUMO (assigned as ππ*) transitions, and the calculated energy gaps are slightly higher for the Rf molecule compared to those of Rz. From a structural aspect, the strong intramolecular hyperconjugative interactions between π → π* orbitals and other feasible intramolecular charge transfer transitions (σ/n → σ*/π*) are characterized through NBO analysis. We also report the global and local reactivity descriptors for the title molecules. The calculated vertical ionization potential and affinity energy values are positive quantities which implies that the molecules mostly get stabilized by gaining electrons. Finally, the NLO properties of the phenoxazin-3-one dyes are also reported. The first-order hyperpolarizability shows the common trend of a linear relation with the molecular dipole moment and an inverse relation with the energy gap. The considerably large β values of the dyes imply that both Rz and Rf molecules might be suitable candidates for designing new organic NLO materials.


 Please wait while we load your content...
Please wait while we load your content...