Removal of Cr(vi) from aqueous solution using magnetic modified biochar derived from raw corncob
Abstract
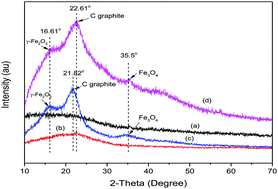
Magnetic modified-corncob biochar with an impregnation ratio of iron at 20% (w/w) was used for removal of Cr(VI) from aqueous solution. Batch adsorption experiments were conducted to investigate the adsorption isotherm, kinetics and mechanism of Cr(VI) onto MCB20. The results indicated that the maximum adsorption capacity achieved was 25.94 mg g−1. The adsorption kinetic data were found to fit best to the pseudo-second order model with a high correlation coefficient (R2 = 0.992). The adsorption mechanisms of Cr(VI) onto MCB20 were electrostatic attraction, anion exchange and adsorption coupled-reduction. The adsorption mechanisms occurring between Cr(VI) anions and MCB20 were due to mainly the contribution of Fe3O4 present in the corncob biochar structure after magnetization by FeCl3. Among the aforementioned mechanisms, adsorption coupled-reduction plays a vital role in removal of Cr(VI) by enhancement of the reduction of Cr(VI) to Cr(III) through generation of electron-donor groups (hydroxyl groups) on the MCB20 surface. Then, Cr(VI) interacted with the electron-donor groups and Cr(VI) is reduced to Cr(III). Besides, Fe2+ ions in the MCB20 structure were simultaneously oxidized to Fe3+ ions and enhanced the transformation of Cr(VI) into Cr(III). The reduced Cr(III) cations, finally, were adsorbed by MCB20 through substitution of Fe3+ with Cr3+ under acidic conditions, complexation with surface functional groups of MCB20 and formation of Cr(OH)3. This study both developed a new way to produce a low cost adsorbent for removal of Cr(VI) from aqueous solution and solved waste by waste.


 Please wait while we load your content...
Please wait while we load your content...