NHC-catalyzed green synthesis of functionalized chromones: DFT mechanistic insights and in vitro activities in cancer cells†
Abstract
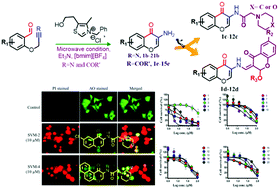
An efficient synthesis of 3-aminochromones and 3-alkylchromones by a N-heterocyclic carbene (NHC) catalyzed intramolecular hydroacylation reaction of corresponding salicylaldehyde derived nitriles and activated alkynes respectively in ionic liquids under microwave conditions is reported. This protocol has the advantages of environmental friendliness, higher yields, shorter reaction times, and convenient operation using the commercially available thiazolium catalyst. The origin of the chemical reactivity of the NHC-catalyzed intramolecular hydroacylation reaction of nitriles is studied using Density Functional Theory (DFT). The results suggest that 3-aminochromone formation occurs via an acyl anion intermediate called a Breslow intermediate (INT2) through TS2. The Breslow intermediate (INT2) forms a carbon–carbon bond with the nitrile carbon to produce an imine intermediate INT3viaTS3, which further undergoes imine to amine tautomerism to give the end product. Some of the derivatives of 3-aminochromone are subjected to amine functionalization in one pot to obtain a library of compounds for anticancer activity. Among the investigated compounds, 2c (SVM-2), 4c (SVM-4) and 2d (SVM-9) show IC50 values of 5.18, 4.89 and 27.3 μM respectively in HeLa S3 cancer cells. Compound 5c (SVM-5) shows IC50 values of 13.3 and 14.2 μM in A549 and HeLa S3 cancer cells, respectively. Compounds 2c (SVM-2) and 4c (SVM-4) produce morphological changes and control the colony formation in HeLa S3 cells, which indicates that these small molecules are potential candidates for anticancer drugs.


 Please wait while we load your content...
Please wait while we load your content...