Experimental evidence for hypercoordination in triorganotelluronium halides with 2-(Me2NCH2)C6H4 groups†‡
Abstract
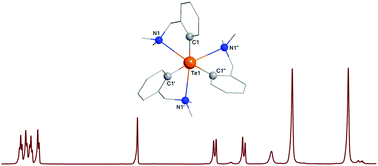
Heteroleptic triorganotelluronium chlorides of the type [RPh2Te]+Cl− (1) and [R2PhTe]+Cl− (4) [R = 2-(Me2NCH2)C6H4] were obtained by reacting Ph2TeCl2 with RLi in 1 : 1 and 1 : 2 molar ratios. The reaction of TeCl4 with RLi (a 1 : 3 molar ratio) afforded a mixture of [R3Te]+Cl− (7) and [R2Te(OH)]+Cl− (10) after separation by column chromatography. Halogen exchange reactions using KX resulted in the corresponding [RPh2Te]+X− [X = Br (2), I (3)], [R2PhTe]+X− [X = Br (5), I (6)] and [R3Te]+X− [X = Br (8), I (9)]. The compounds were characterized by multinuclear NMR spectroscopy and mass spectrometry. The NMR spectra are similar for each series of triorganotelluronium halides (1–3, 4–6 and 7–9, respectively) and no clear evidence for N→Te interactions in solution could be concluded at room temperature, except in the case of compounds 4–6 for which the 1H resonance of the CH2N protons in the pendant arms appears as an AB spin system. For compounds 7 and 10 the low temperature 1H and 13C NMR spectra are consistent with intramolecularly coordinated pendant arms. The solid state structures for compounds 1, 1·0.5CH2Cl2, 3, 4·H2O, 5·H2O, 6, 7·H2O and 10 were determined by single-crystal X-ray diffraction. For all compounds the nitrogen atom of a pendant arm is involved in an intramolecular N→Te interaction in the range 2.571(3)–2.839(8) Å. This results in hypercoordinated triorganotelluronium(IV) cations with different distorted coordination geometries: “see saw” in 1–3, square pyramidal in 4–6 and 10, and octahedral in 7. Further secondary interactions between cations, anions and water molecules were observed in the crystals of these species, resulting in different supramolecular associations.
- This article is part of the themed collection: Selenium & Tellurium chemistry at the beginning of the 3rd millennium: a celebration of ICCST


 Please wait while we load your content...
Please wait while we load your content...