DNA binding, cytotoxic effects and probable targets of an oxindolimine–vanadyl complex as an antitumor agent†
Abstract
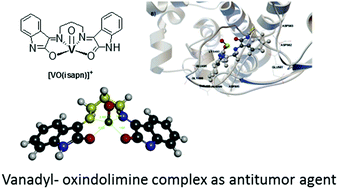
The biological activity of an oxindolimine–oxidovanadium(IV) complex, with significant antiproliferative action against different tumor cells, has been investigated and compared to that of analogous copper(II) or zinc(II) compounds. This vanadyl complex, [VO(isapn)]SO4, was synthesized and characterized by the usual spectroscopic techniques (UV/Vis, IR, and EPR), and its interactions with vital biomolecules were verified. In experiments using a Hoechst 33258 dye bound to DNA, a decrease in the intensity of fluorescence was observed with the increase in the concentration of this complex, revealing its probable binding in grooves of DNA. The determined binding constants indicated a significant change in values with increasing incubation times, which is attributed to probable changes in the metal coordination sphere. However, its influence on the melting point of CT-DNA was negligible, suggesting very little disturbance to the nucleic acid structure. In comparative studies, ΔT = ∼8 °C was observed with the analogous copper(II) complex, [Cu(isapn)]+. In addition, promising results were verified for the cytotoxicity of [VO(isapn)]+ toward human colon and breast cancer cells, with IC50 in the range 5 to 10 μM, after 24 h of treatment at 37 °C. However, the reactivity was much lower against HeLa cells under the same conditions (IC50 > 100 μM). Further studies pointed to topoisomerase IB (Topo I) as a probable additional target of this compound. It inhibited the cleavage step but not the religation, interacting directly with the enzyme and preventing its binding to DNA, similar to what was observed with analogous copper(II) and zinc(II) complexes. Molecular docking simulations showed interactions of the [VO(isapn)]+ complex at different sites of Topo I, corroborating the experimental data.
- This article is part of the themed collection: Vanadium Science: Chemistry, Catalysis, Materials, Biological and Medicinal Studies


 Please wait while we load your content...
Please wait while we load your content...