Facile template synthesis of water-soluble triazine-based Schiff base ligand bridged-coordination polymers of Co(ii), Ni(ii), and Cu(ii): structure, biomolecular interactions, and cytotoxic activity†
Abstract
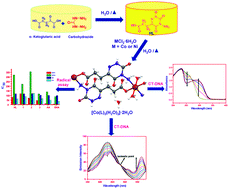
A new set of water-soluble coordination polymers of Co(II), Ni(II), and Cu(II) with 3-(4-amino-3,5-dioxo-2,3,4,5-tetrahydro-[1,2,4]triazin-6-yl)-propionic acid ligand (HL) was synthesized and characterized. The powder X-ray diffraction (PXRD) pattern confirmed the isostructurality between Co(II) and Ni(II) complexes, whereas the Cu(II) complex had a slightly different coordination environment. The cobalt complex has octahedral geometry around the Co(II) ion, at which the triazine ligand (L) was coordinated via an O/N bidentate mode and bridged the two metal centres to form a one-dimensional polymeric unit, which was confirmed by single-crystal X-ray data. In vitro antioxidant assays with DPPH˙ (2,2-diphenyl-1-picrylhydrazyl), ABTS˙+ [2,2′-azinobis-(3-ethylbenzothiazoline-6-sulphonic acid diammonium salt)], NO˙ (nitric oxide), and O2˙− (superoxide) radicals were performed to evaluate the free radical-scavenging ability and showed that the complexes in general and the Cu(II) complex in particular possess significant radical-scavenging activities compared to that of the ligand. The binding affinities towards biomolecules such as calf thymus DNA (CT-DNA) and bovine serum albumin (BSA) were determined, and the copper complex exhibited the highest activity. In addition, the complexes exhibited high cytotoxicity in human breast adenocarcinoma MCF-7 cells in vitro. Morphological changes and apoptosis were assessed using standard thiazolyl blue tetrazolium bromide (MTT) assay, semi-quantitative polymerase chain reaction (PCR) analysis, and acridine orange/ethidium bromide (AO/EB) and DAPI (4′,6-diamidino-2-phenylindole) fluorescence staining assays.


 Please wait while we load your content...
Please wait while we load your content...