Free radical induced selenoxide formation in isomeric organoselenium compounds: the effect of chemical structures on antioxidant activity†
Abstract
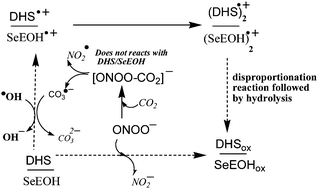
With an aim to understand the effect of structural modifications on antioxidant activity, two structurally isomeric selenium compounds, linear, bis(ethan-2-ol)selenide (SeEOH), and cyclic, DL-trans-3,4-dihydroxy-1-selenolane (DHS), were compared. The antioxidant activity was assigned through their ability to scavenge reactive oxygen species (ROS), where both compounds indicated formation of selenoxides. The ROS identified for the present study are peroxynitrite, hydroxyl (˙OH) and carbonate (CO3˙−) radicals. The rate constant for the scavenging of both peroxynitrite and the CO3˙− radical was higher for DHS as compared to that for SeEOH. The one-electron oxidation during these reactions indicated the formation of a selenium centred dimer radical cation (![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Se∴Se
Se∴Se![[double bond splayed right]](https://www.rsc.org/images/entities/char_e00a.gif) )+, as the intermediate, which undergoes the disproportionation reaction to form the corresponding selenoxide. Quantitative estimation of selenoxides indicated that cyclic compound DHS produced a higher yield of selenoxides than the linear compound SeEOH due to higher stability of (
)+, as the intermediate, which undergoes the disproportionation reaction to form the corresponding selenoxide. Quantitative estimation of selenoxides indicated that cyclic compound DHS produced a higher yield of selenoxides than the linear compound SeEOH due to higher stability of (![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) Se∴Se
Se∴Se![[double bond splayed right]](https://www.rsc.org/images/entities/char_e00a.gif) )+. The selenoxides undergo reduction by thiols, thereby regenerating back to the parent compound. The results thereby suggest that formation of selenoxides improves the antioxidant activity of organoselenium compounds and should be considered as an important step in the design of new selenium based antioxidants.
)+. The selenoxides undergo reduction by thiols, thereby regenerating back to the parent compound. The results thereby suggest that formation of selenoxides improves the antioxidant activity of organoselenium compounds and should be considered as an important step in the design of new selenium based antioxidants.
- This article is part of the themed collection: Selenium & Tellurium chemistry at the beginning of the 3rd millennium: a celebration of ICCST


 Please wait while we load your content...
Please wait while we load your content...