Evaluation of silver-incorporating zeolites as bifunctional electrocatalysts for direct borohydride fuel cells
Abstract
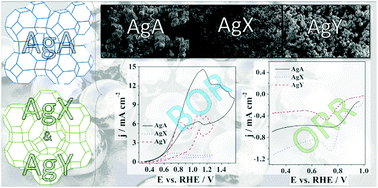
Three zeolites (A, X and Y) with Ag incorporated in their cages are tested as bifunctional electrocatalysts for direct borohydride fuel cells (DBFCs). Ag zeolites are characterised by XRD, FTIR, nitrogen sorption and SEM, as well as by electrochemical methods. Ag zeolite shows good activity for the anodic reaction, i.e., borohydride oxidation reaction (BOR) in alkaline media, presenting low onset potential and high current densities. BOR kinetic and faradaic parameters at AgY are calculated, with the number of exchanged electrons being 4.3. The BOR at AgY was found to be of 0.6 order with respect to BH4− concentration with an activation energy of 13 kJ mol−1. Regarding ORR, AgX shows the best performance with the highest current densities and good stability over time. A two-electron pathway is observed for ORR at AgX and specific capacitance was calculated to be 5.61 F g−1. The DBFC performance is investigated based on half-cell measurements of the AgY anode and Pt/C cathode.


 Please wait while we load your content...
Please wait while we load your content...