Synthesis and anticancer cytotoxicity with structural context of an α-hydroxyphosphonate based compound library derived from substituted benzaldehydes†
Abstract
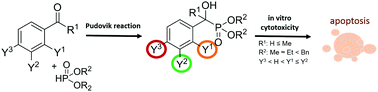
We synthesized substituted benzaldehyde derived α-hydroxyphosphonates (αOHP), α-hydroxyphosphonic acids (αOHPA) and α-phosphinoyloxyphosphonates (αOPP) and characterized their cytotoxicity against a panel of cancer cell lines. A library containing 56 analogues was screened against Mes-Sa parental and Mes-Sa/Dx5 multidrug resistant uterine sarcoma cell lines, using a fluorescence-based cytotoxicity assay. The cytotoxicity screening revealed that dibenzyl-αOHPs and dimethyl-α-diphenyl-OPPs were the most active clusters, which encouraged us to synthesize further dibenzyl-α-diphenyl-OPP derivatives that elicited pronounced cell killing. Further structure–activity relationships showed the relevance of hydrophobicity and the position of substituents on the main benzene ring as determinants of toxicity. The most active analogs proved to be equally, or even more toxic to the multidrug resistant (MDR) cell line Mes-Sa/Dx5, suggesting these compounds may overcome P-glycoprotein mediated multidrug resistance by evading the drug transporter.


 Please wait while we load your content...
Please wait while we load your content...