In situ synthesized hollow spheres of a silica–ruthenium–nickel composite catalyst for the hydrolytic dehydrogenation of ammonia borane
Abstract
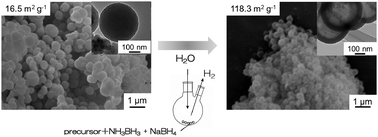
The present study investigated the fabrication of hollow spheres of a silica–ruthenium–nickel composite catalyst for the hydrolytic dehydrogenation of ammonia borane. The hollow spheres were obtained via the originally developed in situ synthesis method. In this method, the hollow spheres were formed by treating in an aqueous solution, including sodium borohydride and ammonia borane, from spherical silica–nickel–ruthenium composite particles prepared via a sol–gel method. In this process, the formation of hollow voids, the activation of active ruthenium and nickel species and the hydrolysis of ammonia borane for hydrogen evolution occurred parallely. Compared to the catalytic activity of hollow spheres with various ruthenium/(ruthenium + nickel) ratios and to the total amount of metals, the hollow spheres with a ruthenium/(ruthenium + nickel) ratio = 0.5 and a total amount of metals = 8.0 mol% showed the highest activity for the hydrogen evolution with a comparative turnover frequency (33.6 mol-H2 min−1 [mol-(Ru + Ni)]−1) of the previously reported catalyst. The hollow spheres included spherical particles with a diameter of the hollow void = ca. 250 nm, and their specific surface area was about ten times as high as the spherical particle precursor, indicating that the obtained hollow spheres consisted of a porous structure. The highly active hollow spheres showed cycle ability for hydrogen evolution and their activity did not significantly decrease with the increase in the cycle number.


 Please wait while we load your content...
Please wait while we load your content...