Porous surface of an achiral trimer in the chiral conglomerate phase catalyzes a direct aldol reaction†
Abstract
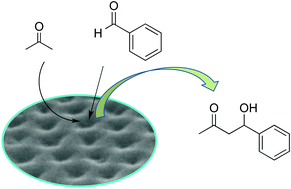
A flexible liquid-crystalline trimer in the optically isotropic chiral conglomerate phase catalyzes the direct aldol reaction of acetone with benzaldehyde. We report that the trimer exhibiting a porous surface acts as an enzyme-mimetic catalyst.


 Please wait while we load your content...
Please wait while we load your content...