Synthesis of fisetin co-crystals with caffeine and nicotinamide using the cooling crystallization technique: biopharmaceutical studies
Abstract
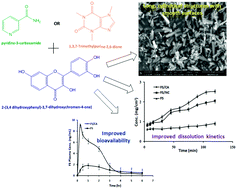
Fisetin (FS), 2-(3,4 dihydroxyphenyl)-3,7-dihydroxychromen-4-one), a bioactive natural hydrophobic flavonol, has numerous therapeutic properties. However, its efficacy as a potential phytochemical has been limited owing to poor aqueous solubility and consecutively bioavailability. In the present study, we report the co-crystal synthesis of FS with caffeine (CA, 1,3,7-trimethylpurine-2,6-dione) and a nicotinamide (NC, pyridine-3-carboxamide) co-former using cooling crystallization technology to augment the biopharmaceutical properties of FS. A methanolic solution containing FS and co-formers was subjected to cooling crystallization to acquire the FS co-crystals. The developed co-crystals were analyzed using differential scanning calorimetry, Fourier transform infrared spectroscopy, X-ray diffractometry, scanning electron microscopy, saturation solubility and intrinsic dissolution studies. The oral bioavailability of the co-crystals was studied using healthy Wister male rats. In addition, the anti-inflammatory potential of the co-crystals was studied using a carrageenan-induced rat paw edema model. Cooling crystallization caused development of golden yellow colored cylindrical FS–CA and rectangular FS–NC co-crystals, as verified by thermal and diffractometry analysis which exhibited more than a two-fold enrichment in the solubility of FS. The FS–CA co-crystal displayed a marked three-fold improvement in the oral bioavailability of FS and retained the anti-inflammatory potential of FS in the pharmacodynamic study. The investigation showed the utility of cooling crystallization technology to augment the biopharmaceutical properties of bioactive FS and could be explored for clinically important lipophilic phytochemicals to improve their pharmaceutical properties.


 Please wait while we load your content...
Please wait while we load your content...