Efficient selective 4-aminophenol sensing and antibacterial activity of ternary Ag2O3·SnO2·Cr2O3 nanoparticles†
Abstract
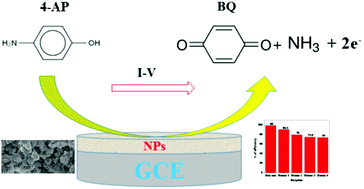
In this study, Ag2O3·SnO2·Cr2O3 nanoparticles (NPs) were synthesized via a simple and quick co-precipitation method. The produced materials were characterized by X-ray powder diffraction (XRD), scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), X-ray photoelectron spectroscopy (XPS), Fourier-transform infrared spectroscopy (FTIR), and photoluminescence (PL) spectroscopy. The hydrodynamic size and zeta potential were measured under acidic and basic pH conditions. The photocatalytic activities of the newly synthesized composite were studied considering various parameters. Antibacterial activity tests showed that Ag2O3·SnO2·Cr2O3 is an excellent anti-bacterial agent. A reliable electrochemical sensor probe was designed by depositing the synthesized Ag2O3·SnO2·Cr2O3 NPs onto the flat part of GCE, which showed excellent oxidation activity and selectivity towards 4-aminophenol (4-AP) in a phosphate buffer medium. During electrochemical analysis, the desired 4-AP sensor responded linearly over a large concentration range (0.1 nM–0.01 mM), which is known as the linear dynamic range (LDR), and exhibited appreciable sensitivity (25.1962 μA μM−1 cm−2). Furthermore, it showed a considerably low limit of detection (98.41 ± 4.92 pM), good reproducibility, fast response (20.0 s), and long-term stability in the phosphate buffer medium. This is an efficient method for the fabrication of a selective electrochemical sensor using the electrochemical approach for safety in environmental and healthcare fields.


 Please wait while we load your content...
Please wait while we load your content...