A three-dimensional nickel–cobalt oxide nanomaterial as an enzyme-mimetic electrocatalyst for the glucose and lactic acid oxidation reaction†
Abstract
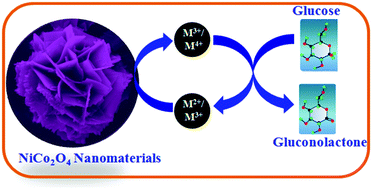
Here we demonstrate a highly porous three-dimensional nickel–cobalt oxide (NiCo2O4) nanomaterial as a potential glucose oxidase (GOx) enzyme-mimicking catalyst for the electrochemical oxidation of glucose and lactic acid in alkaline medium. The as-synthesized NiCo2O4 nanomaterial based anode exhibits an excellent catalytic activity towards glucose oxidation with a low oxidation potential of ∼0.43 V (vs. Ag/AgCl), a high current density of ∼26.2 mA cm−2, a high mass activity of ∼263.0 A g−1 and a long term durability of over ∼500 continuous cycles. The flower-like NiCo2O4 nanomaterial delivers an over ∼1.4, ∼1.9, and ∼2.0 times higher catalytic current density with a less positive shift by ∼70 mV, ∼80 mV and ∼40 mV, respectively, when compared to NiO, Co3O4, and commercial Pt/C. The enhanced catalytic activity of the as-developed Earth-abundant NiCo2O4 nanomaterial may be ascribed to its exclusive architecture with a less agglomerated structure, high electrochemically active surface area (ECASA), small pore size, mixed active sites of nickel and cobalt cations and favourable atmosphere for the enriched electron transfer process between analytes and electrodes. The present anode material is competent in performing direct electron transfer to the surface of the electrode and removes the requirement for enzymes and mediators.


 Please wait while we load your content...
Please wait while we load your content...