Detection of uric acid based on doped ZnO/Ag2O/Co3O4 nanoparticle loaded glassy carbon electrode†
Abstract
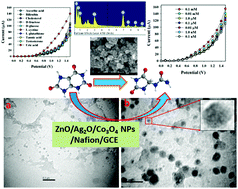
A highly sensitive uric acid (UA) sensor was fabricated using a wet-chemical (co-precipitation) method to prepare doped ZnO/Ag2O/Co3O4 nanoparticles (NPs) and load them onto a glassy carbon electrode (GCE) by an electrochemical approach. The detailed characterization of the NPs was performed by using conventional methods, such as X-ray photoelectron spectroscopy (XPS), ultraviolet-visible spectroscopy (UV-vis.), Fourier-transform infrared spectroscopy (FTIR), field emission scanning electron microscopy (FESEM), Tunneling electron microscopy (TEM), energy-dispersive X-ray spectroscopy (EDS), and X-ray powder diffraction (XRD) analysis. Thermal gravimetric analysis (TGA) of the as-prepared ternary NPs was performed in order to study the stability of NPs in different temperature ranges over which the weight loss and thermal effect are significant. During the electrochemical analysis, the proposed UA sensor was found to be linear over a large linear dynamic range (LRD; 0.1 nM–0.01 mM). The analytical performance of the sensor such as sensitivity (82.3323 μA μM−1 cm−2) was estimated from the slope of the calibration curve and the detection limit (89.14 ± 4.46 pM) was calculated at a signal to noise ratio of 3. The proposed UA biosensor showed reliable reproducibility, a short response time (22.0 s), long-term stability, and no interference effects. The ZnO/Ag2O/Co3O4 NPs/GCE sensor was also validated with real biological samples. Thus, this method might be a prospective and reliable method for the future development of enzyme-free biosensors using doped ternary metal oxides in broad scales.


 Please wait while we load your content...
Please wait while we load your content...