Thiol-/thioether-functionalized porous organic polymers for simultaneous removal of mercury(ii) ion and aromatic pollutants in water†
Abstract
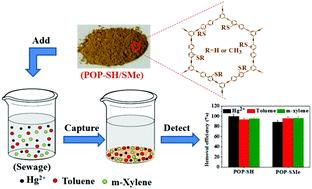
The purpose of this work is to prepare effective adsorbents for simultaneously removing Hg(II) ion and aromatic pollutants in water, which still remains a great challenge presently due to their different physicochemical properties. Herein, two new thiol-/thioether-functionalized porous organic polymers were prepared and characterized by scanning electron microscopy, infrared spectra, 13C CP/MAS nuclear magnetic resonance spectra, energy-dispersive X-ray spectroscopy, elemental analysis, thermo-gravimetric analysis, and nitrogen adsorption–desorption isotherms. The results showed that the two adsorbents had a loosely porous structure, high BET surface area, and good thermal and chemical stability. The optimal pH value for the two new adsorbents to uptake Hg(II) was 3–4. The new adsorbents presented a high adsorption ability with the maximum adsorption capacity of 180 mg g−1 for Hg(II) and 358–452 mg g−1 for aromatic pollutants (toluene and m-xylene as models) and acceptable/fast binding kinetics for Hg(II) and aromatic pollutants, respectively. The adsorbents also showed high adsorption selectivity for Hg(II) in the presence of commonly coexisting metal ions. Moreover, the two adsorbents had good simultaneous removal ability for Hg(II) and the aromatic pollutants at different concentrations and good reusability. Finally, the two new adsorbents were used successfully for the simultaneous and highly efficient removal of Hg(II) ion and aromatic pollutants in simulated sewage with removal efficiencies higher than 88% for Hg(II) and higher than 93% for the aromatic pollutants (10 mg of adsorbent mixed with 10 mL of sewage containing Hg(II) and the aromatic pollutants at 10 μg mL−1 for each one), indicating their great potential to be applied for the simultaneous removal of Hg(II) and aromatic pollutants in real sewage or wastewater.


 Please wait while we load your content...
Please wait while we load your content...