Hydrogen bonded dimers of ketocoumarin in the solid state and alcohol:water binary solvent: fluorescence spectroscopy, crystal structure and DFT investigation†
Abstract
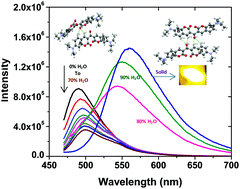
The photophysical properties of 3,3′-carbonylbis(7-diethylamino)-2H-chromen-2-one (ketocoumarin) were investigated in the solid state, different solvents and an alcohol:water binary mixture using steady state, time-resolved fluorescence spectroscopy, XRD studies and DFT calculations. In neat solvents ketocoumarin (KC) shows a small Stokes shift as compared to other 7-diethylaminocoumarin (7-DEC) dyes and this is due to the π–π* electronic transitions involving the bridge keto group. In aqueous alcoholic solution, the fluorescence maximum of KC depends on the percentage of water and a new emission maximum at 550 nm with a large Stokes shift is observed at 90% water content. The solid state fluorescence and XRD crystal structure analysis reveals that the new decay component with a lifetime of 2.71 ns and new emission maximum in the alcohol:water mixture is due to the intermolecular hydrogen bonding [HB] interaction between KC molecules in the alcohol:water binary mixture. The XRD structure reveals the two modes of intermolecular CH⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C hydrogen bonding (HB) which lead to two dimeric forms of KC, D1 and D2. The emission wavelength dependent excitation spectrum in the solid state and alcohol:water binary mixture confirms the H-type nature of dimer D1 and the J-type nature of dimer D2. The time-resolved fluorescence studies indicate that the new decay component with a lifetime of 2.71 ns is due to dimer D1 and the new emission with the maximum at 550 nm is due to dimer D2. Density functional theory (DFT) analysis confirms that the KC dimers are stabilized by hydrogen bonding and other non-covalent interactions. The hydrogen bonded KC dimers are responsible for the anomalous fluorescence behavior of KC in the alcohol:water binary mixture and for the bright yellow fluorescence in the solid state.
C hydrogen bonding (HB) which lead to two dimeric forms of KC, D1 and D2. The emission wavelength dependent excitation spectrum in the solid state and alcohol:water binary mixture confirms the H-type nature of dimer D1 and the J-type nature of dimer D2. The time-resolved fluorescence studies indicate that the new decay component with a lifetime of 2.71 ns is due to dimer D1 and the new emission with the maximum at 550 nm is due to dimer D2. Density functional theory (DFT) analysis confirms that the KC dimers are stabilized by hydrogen bonding and other non-covalent interactions. The hydrogen bonded KC dimers are responsible for the anomalous fluorescence behavior of KC in the alcohol:water binary mixture and for the bright yellow fluorescence in the solid state.


 Please wait while we load your content...
Please wait while we load your content...