Adaptable sensor for employing fluorometric detection of methanol molecules: theoretical aspects and DNA binding studies†
Abstract
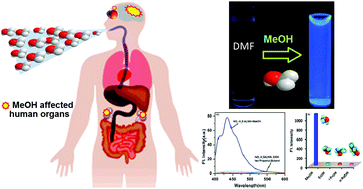
The multifunctional Schiff base ligand N,N′-bis(5-nitro-salicylaldehyde)azine (NO2-H2SALNN) has been successfully synthesized and characterized by ESI-MS+, 1H-NMR spectroscopy and CHN elemental analysis. The very feeble intrinsic fluorescence exerted by the ligand was found to be enhanced by several folds in the presence of methanol (∼168 fold) and water (∼10 fold) molecules. This enhancement of fluorescence intensity is supposed to occur as a result of blocking of rotational isomerization along the azomethine group (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) due to its coordination with solvent molecules, thereby causing turn on fluorescence at two distinct wavelengths in the presence of methanol and water having a large difference between the two emission wavelengths (Δλ = 125 nm). The hydrogen bond-assisted enhancement fluorescence (HAEF) for methanol and water at two different wavelengths giving two different emissions may be due to the different size/nature of the solvents as well as differences in solvent polarity. The binding of NO2-H2SALNN and the solvent molecules was investigated and described by spectroscopic and computational studies. The bioactivity of NO2-H2SALNN has also been inspected by DNA binding measurements through spectrometric and thermodynamic studies. DNA binding studies reveal that the ligand interacts with double stranded CT-DNA through a groove binding mode and the intrinsic binding constant was determined by calorimetric studies to be (2.24 ± 0.04) × 105 M−1.
N) due to its coordination with solvent molecules, thereby causing turn on fluorescence at two distinct wavelengths in the presence of methanol and water having a large difference between the two emission wavelengths (Δλ = 125 nm). The hydrogen bond-assisted enhancement fluorescence (HAEF) for methanol and water at two different wavelengths giving two different emissions may be due to the different size/nature of the solvents as well as differences in solvent polarity. The binding of NO2-H2SALNN and the solvent molecules was investigated and described by spectroscopic and computational studies. The bioactivity of NO2-H2SALNN has also been inspected by DNA binding measurements through spectrometric and thermodynamic studies. DNA binding studies reveal that the ligand interacts with double stranded CT-DNA through a groove binding mode and the intrinsic binding constant was determined by calorimetric studies to be (2.24 ± 0.04) × 105 M−1.


 Please wait while we load your content...
Please wait while we load your content...