Zirconium-MOF-catalysed selective synthesis of α-hydroxyamide via the transfer hydrogenation of α-ketoamide†
Abstract
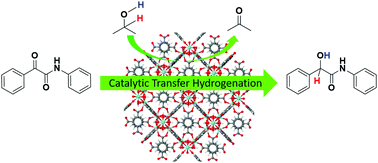
This work reports the synthesis of α-hydroxy amide and its derivatives using zirconium-based metal–organic frameworks (Zr-MOFs). The Zr-MOF was prepared using a ligand containing different functionalities as a linker with different porosities. The catalyst efficiently facilitated the transfer hydrogenation of α-ketoamide to α-hydroxyamide. The reaction involved a green hydrogen source, namely isopropyl alcohol, which also acted as a solvent. The role of the ligand in the catalyst was optimized for the selective conversion of α-ketoamide to α-hydroxyamide. UiO-66 (Zr) crystal was efficiently used for the first time for the hydrogenation of α-ketoamide to α-hydroxyamide. The catalyst was recovered and recycled five times without any loss in activity and selectivity performance. The morphology, activity and stability of the UiO-66 (Zr) catalyst were analyzed using field emission gun scanning electron microscopy (FEG-SEM), X-ray diffraction (XRD), infra-red (IR) spectroscopy and thermogravimetric analysis (TGA). The existence of α-hydroxyamide and its derivatives was confirmed from 1H and 13C-NMR.


 Please wait while we load your content...
Please wait while we load your content...