Excited-state intramolecular proton transfer (ESIPT) emitters based on a 2-(2′-hydroxybenzofuranyl)benzoxazole (HBBO) scaffold functionalised with oligo(ethylene glycol) (OEG) chains†
Abstract
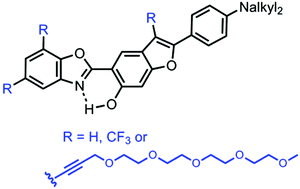
This article describes the multi-step synthesis of 2-(2′-hydroxybenzofuran)benzoxazole (HBBO) derivatives functionalised with one to three oligo(ethylene glycol) (OEG) chains with the goal to allow a good vectorization in aqueous media. Benzoxazole dyes are well-known organic fluorophores exhibiting excited-state intramolecular proton transfer (ESIPT) emission. The insertion of these highly hydrophilic OEG chains allows a good solubilization of these dyes in a wide range of solvents of different polarity including PBS buffer/DMSO (95/5) and water, for one of them. The photophysical properties of these ESIPT emitters in solution, and also in the solid-state, as doped as 1 wt% in poly(methylmethacrylate) (PMMA) or polystyrene (PS) films, are discussed. Specifically, the presence of an OEG chain at the 3-position of the benzofuran ring was found to sizeably enhance the fluorescence intensity of the ESIPT emission in the solution state.


 Please wait while we load your content...
Please wait while we load your content...