Synthesis, in vitro COX-1/COX-2 inhibition testing and molecular docking study of novel 1,4-benzoxazine derivatives†
Abstract
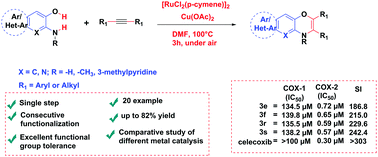
The present work deals with an efficient and straightforward synthesis, biological activity and molecular docking study of novel 1,4-benzoxazine derivatives. Dehydrocyclization through consecutive functionalization of the –OH and –NH bonds of 2-aminophenol derivatives with disubstituted alkynes in the presence of [RuCl2(p-cymene)]2 as a catalyst allows synthesis of 1,4-benzoxazine derivatives in a single synthetic step. Inspired by a literature survey of COX-1/COX-2 enzyme inhibition studies we found that the biphenyl type structure is more suitable to inhibit the concentration of COX-/COX-2 enzymes, and to check the viability of this assumption we performed molecular docking as well as an in vitro COX-1/COX-2 inhibition study for the synthesised derivatives. The results of the assay indicated that compounds 3e, 3f, 3r, and 3s exhibited optimal COX-2 inhibition, with IC50 values of 0.57–0.72 μM, and the selectivity index (SI) range of 186.8–242.4, using Celecoxib as a standard drug (IC50 = 0.30 μM; COX-2 SI: >303). Furthermore, the molecular docking study explored the essential binding mode and possible interactions within the binding site of COX-1/COX-2 isoenzymes. This study identified potential compounds, which can be promising leads for their subsequent development as newer anti-inflammatory agents.


 Please wait while we load your content...
Please wait while we load your content...