Thermally oxidized CdS as a photoactive material†
Abstract
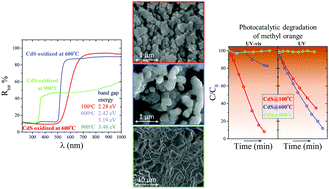
A cadmium sulfide nanopowder was precipitated from water-based precursors and oxidized in air at temperatures ranging from 100 to 900 °C. CdS and the oxidized powders were examined by means of XRD, SEM, EDX, DTA/TG, spectrophotometry, and photocatalytic tests. XRD analysis revealed that CdS had crystallized as a mixture of cubic and hexagonal polymorphs and no changes in its composition were found below 400 °C. At higher temperatures, different oxidized phases belonging to the Cd–O–S system is observed. The appearances of Cd(S2O7) and CdSO4 at 400 °C is accompanied by an increase in the crystallinity. Cd5S3O6, Cd3SO6, and CdO appear at 500, 600, and 800 °C, respectively. SEM observations showed that oxidation leads to a gradual increase in the size of the CdS nanoparticles from 10 nm to more than 3.5 μm at 800 °C. The fundamental absorption edge is present in the visible range of light up to 700 °C, accompanied by the edge in the UV range, which appears at 600 °C. Based on the zeta potential measurements a representative of anionic dyes – methyl orange, was chosen for the photocatalytic experiments under UV and UV-vis light. For the first time, the incredible photoactivity of the oxidized CdS was demonstrated. It was shown that both Cd5S3O6 and Cd3SO6 yield unique photoactivities in the UV range.


 Please wait while we load your content...
Please wait while we load your content...