Unusual complexation behaviors of R-BTPs with water molecule and Pd(ii) caused by electronic modulation of substituents on BTP backbone: new insights into palladium separation under the framework of minor actinides’ partitioning†
Abstract
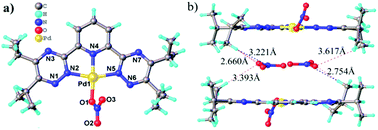
2,6-Bis(5,6-dialkyl-1,2,4-triazin-3-yl)-pyridine (R-BTP) is one of the most promising classes of soft N-donor ligands for minor trivalent actinide (such as Am(III) and Cm(III)) separation but its complexation behavior with interfering element Pd(II) is still unknown. The effect of substituents (R-) on the modulation of R-BTP's complexation with Pd(II) was studied by single crystal diffractometry, 1H NMR spectroscopy, ESI-MS, and density functional theory (DFT) calculation methods. The formation of both 1 : 1 and 2 : 1 complexes of R-BTP with Pd(II) were firstly identified, and their electronic and geometric structures were determined. It has been found that the modification of the R-BTP skeleton with substituents (R-) changing from methyl (C1) to s-butyl (C4) leads to two different coordination models with water. The structures of 2 : 1 R-BTP/Pd(II) complexes were determined to be unusually paramagnetic, distorted six-coordinate compounds. However, the 1 : 1 species was found to have a typical square-planar geometry. The extraction properties of four R-BTPs for Pd(II) and other fission or corrosion products were studied. This work can help to further understand the complexation behavior of Pd(II) with R-BTP, which provides theoretical insights into the partitioning of minor actinides and Pd(II) from high level active waste (HLW) using R-BTP type ligands.


 Please wait while we load your content...
Please wait while we load your content...