First report of silver ion recognition via a silatrane-based receptor: excellent selectivity, low detection limit and good applicability†
Abstract
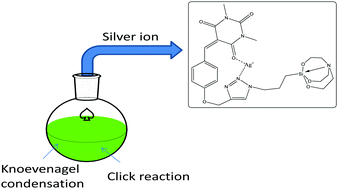
The development of selective and sensitive complex molecular systems for the detection of Ag+ ions is of great importance because the extensive use of the toxic silver containing materials has adverse effects on the human health and the environment. Here, we report a new chemosensor for silver ion recognition in aqueous solutions, which was constructed by integrating three moieties: silatrane, 1,2,3-triazole and barbituric acid, using Knoevenagel condensation and Cu(I) catalyzed [2+3] azide–alkyne cycloaddition reaction. The sensing assay based on silatrane exhibited high selectivity and high specificity towards silver ions. Overall, the results show that this strategy to develop new functional materials can be employed for the efficient sensing of Ag+ ions. Further, the benefit of the existence of silicon in the receptor lies in the extension of this molecular system in the field of nanochemistry, where silica-based nanomaterials can have more significant outcomes. Moreover, the ion detection ability in aqueous solution makes the sensor suitable to be employed for detection in real samples.


 Please wait while we load your content...
Please wait while we load your content...