Contribution to the coordination chemistry of transition metal nitroprussides: a cryo-XPS study†
Abstract
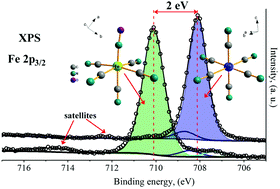
The series of coordination polymers under investigation was formed by the assembly of a pentacyanonitrosylferrate(II) anionic block, [Fe(CN)5NO]2−, through monovalent and divalent transition metal ions, e.g. Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+, Hg2+ and Ag+. For divalent ions, the resulting materials have a 3D porous framework with attractive features for applications in gas storage and separation, as electroactive solids, light-driven molecular magnets, and so on; in this study, we report the results obtained for a series of coordination polymers using the cryogenic X-ray photoelectron spectroscopy (cryo-XPS) data; comprehensive details regarding their coordination chemistries were obtained from the acquired spectra in addition to their comparison with the structural and spectroscopic information obtained from other techniques. The results discussed herein are original and contribute towards the understanding of the electronic structures and related properties for this family of coordination polymers. This series of solids was found to be highly susceptible to strong damage induced by X-ray beams throughout the conventional XPS experiment; therefore, the analysis was conducted under cryogenic conditions.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...