Toxic organic solvent adsorption by a hydrophobic covalent polymer†
Abstract
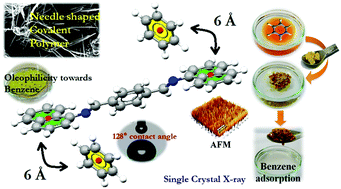
A low cost easy to construct imine-based covalent polymer (CPCMERI-1) has been successfully prepared via a room temperature Schiff-base condensation reaction. Purposeful use of an electron rich naphthalene moiety as a skeletal unit implants hydrophobicity in the polymer. The contact angle of the hydrophobic covalent polymer coated surface has been found to be as high as 128°. The newly developed CPCMERI-1 has high thermal as well as chemical stability due to the presence of extended π–π conjugation of naphthalene and phenyl rings linked through –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– bonds. Taking advantage of the high stability factor and hydrophobicities of the covalent polymer, we have performed liquid phase adsorption of benzene and its electron rich, and electron deficient substituents like toluene and nitrobenzene, respectively. CPCMERI-1 selectively adsorbs benzene although it possesses adsorption affinity towards nitrobenzene and toluene. The strong π–π interaction between the benzene and naphthalene moiety of the polymer is responsible for the adsorption. Surface analysis of CPCMERI-1 before and after adsorption of benzene has been investigated which depicts the surface adsorption phenomena of the analyte. The interaction has been further examined by using DFT-D3 study which reveals that benzene interacts with the host napthalene moiety through π–π interaction. Precise experimentation reveals that CPCMERI-1 exhibits better performance than commercially available activated carbon and some recently reported materials towards adsorption of organic solvents like benzene, toluene, nitrobenzene etc. We expect that our work presented here will herald a new type of polymer as CPCMERI-1 may be a useful adsorbent for removing organic pollutants.
N– bonds. Taking advantage of the high stability factor and hydrophobicities of the covalent polymer, we have performed liquid phase adsorption of benzene and its electron rich, and electron deficient substituents like toluene and nitrobenzene, respectively. CPCMERI-1 selectively adsorbs benzene although it possesses adsorption affinity towards nitrobenzene and toluene. The strong π–π interaction between the benzene and naphthalene moiety of the polymer is responsible for the adsorption. Surface analysis of CPCMERI-1 before and after adsorption of benzene has been investigated which depicts the surface adsorption phenomena of the analyte. The interaction has been further examined by using DFT-D3 study which reveals that benzene interacts with the host napthalene moiety through π–π interaction. Precise experimentation reveals that CPCMERI-1 exhibits better performance than commercially available activated carbon and some recently reported materials towards adsorption of organic solvents like benzene, toluene, nitrobenzene etc. We expect that our work presented here will herald a new type of polymer as CPCMERI-1 may be a useful adsorbent for removing organic pollutants.


 Please wait while we load your content...
Please wait while we load your content...