Structural dependence of the optical properties of narrow band gap thiophene–thiadiazoloquinoxaline derivatives and their application in organic photovoltaic cells†
Abstract
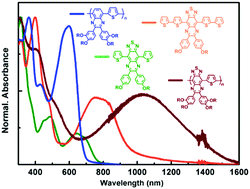
Two new molecules, TQT1 and TQT2, and a polymer, PTQT, with a core based on [1,2,5]thiadiazolo[3,4-g]quinoxaline (TQ) with oligoether-substituted phenyl groups as acceptor and thiophene (T) or bithiophene (T2) as donor, were synthesised, characterised, and used in photovoltaic devices. An analogous polymer, PQT, with quinoxaline units (Q) replacing TQ was also synthesised for comparison purposes. The optical band gap decreases in the order: PQT (1.83 eV), TQT1 (1.68 eV), TQT2 (1.31 eV), PTQT (0.78 eV), this being one of the smallest optical band gaps obtained for conjugated polymers. Density functional theory calculations along with solvatochromic studies demonstrated that in TQT1 the conjugation extension brought about by the thiophene rings affects mainly the HOMO orbital, leaving the LUMO localized in the TQ unit. In consequence, HOMO–LUMO excitations have intramolecular charge transfer (ICT) character from the thiophene ends to the TQ core and the addition of extra thiophene units (TQT2) induces a decrease in the optical gap. The comparison with the polymer PTQT further elucidates on the role of delocalization extension to the observed shift of the lower energy transitions to the near infrared (NIR) spectral range. All the materials were used as electron-donors in solution-processed bulk heterojunction solar cells (BHJ-OSCs) with PCBM as acceptor and TQT1 was also used as electron-acceptor upon combination with the polymer PTB7, thereby revealing its ambipolar behavior. The spectral response of the cells incorporating the materials with the lowest band gaps extended beyond the longest wavelength of illumination tested (800 nm). The device performances are modest but the open-circuit voltages (VOC) can be well correlated with the optical band gaps.


 Please wait while we load your content...
Please wait while we load your content...