Synthesis, characterization and photophysical studies of a novel polycyclic diborane†
Abstract
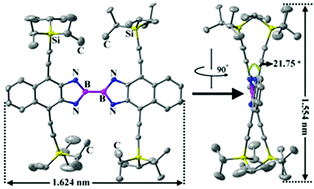
Treatment of tetrakis(dimethylamino)diboron (B2(NMe2)4) with 1,4-bis(2-(triisopropylsilyl)ethynyl)naphthalene-2,3-diamine (1) generated a large NBBN polycyclic compound 1,1-B2{2,3-(NH)2-1,4-(iPr3SiC2)2C10H4} (2). The X-ray structure analysis reveals that 2 has a twisted structure (dihedral angle ≈ 21.75°) with a rather short B–B bond (1.667(9) Å). Theoretical calculations on 1,1-B2 isomer 2 and its counterpart 1,2-B2 isomer 3 have been conducted to understand their frontier orbital energy levels. Compound 2 shows strong emission with a high quantum yield of 0.52, despite the presence of many unbeneficial non-radiative behaviors such as the vibration of the TIPS groups and the rotation of the B–B single bond.


 Please wait while we load your content...
Please wait while we load your content...