NHC stabilized Pd nanoclusters in the Mizoroki–Heck reaction within microemulsion: exploring the role of imidazolium salt in rate enhancement†
Abstract
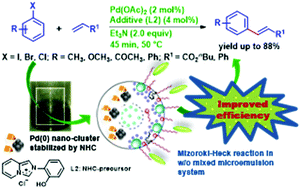
We report the significant rate enhancement of the Mizoroki–Heck reaction by in situ generated palladium nanoclusters within the confined space of water-in-oil (w/o) mixed microemulsion (μE) formulated by sodium dodecylsulfate (SDS), polyoxyethylene (23) lauryl ether (Brij-35) and isopropyl myristate (IPM), in the presence of novel imidazo[1,5-α]pyridinium chlorides as N-heterocyclic carbene (NHC) precursors. This is the first endeavor to study an organic reaction in microemulsions containing IPM as a biocompatible green oil. A series of novel imidazo[1,5-α]pyridinium chlorides were synthesized in a cost effective manner and were screened for a typical Heck reaction between 4-iodotoluene and nbutylacrylate. The optimum rate was achieved with 2-hydroxyphenylimidazo[1,5-α]pyridinium chloride at low catalyst loading under mild conditions. Conductivity and FTIR measurements suggested an effective change in the hydrogen bonding network of interfacial water in the presence of the NHC-precursor. Strong evidence for in situ formation of the Pd–NHC complex was obtained which significantly accounts for the rate enhancement. The reaction progresses rapidly with non-activated and sterically hindered aryl halides under optimal conditions. We further report a unique isolation technique to separate IPM (oil) from the reaction medium. The effect of NHC-precursors within the confined space of a microemulsion, wherein the reaction takes place, is correlated with the observed rate enhancement.


 Please wait while we load your content...
Please wait while we load your content...