Improved catalytic performance of lipase under non-aqueous conditions by entrapment into alkyl-functionalized mesoporous silica†
Abstract
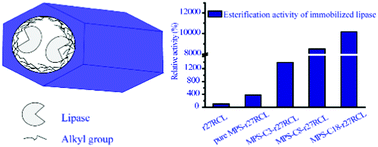
Three alkyl-functionalized mesoporous silica materials with different lengths of hydrocarbon chains (propyl, octyl and octadecyl) were synthesized by a one-pot condensation approach to enhance the catalytic performance of immobilized lipase r27RCL under non-aqueous conditions. The catalytic performance (esterification activity, thermal stability in n-hexane and organic solvent tolerance) of immobilized r27RCL greatly increased with the enhanced surface hydrophobicity of the mesoporous silica materials, mainly because of lipase activation via hydrophobic interactions between alkyl groups in the mesoporous silica materials and the surface loop (so-called “lid”) of lipase r27RCL. Furthermore, lipase r27RCL immobilized into mesoporous silica materials modified with octadecyl showed remarkably increased activity for the esterification reaction between ethanol and lauric acid (∼101 times higher than that of free lipase) and enantioselectivity in the kinetic resolution of (R, S)-1-phenylethanol by vinyl acetate in hexane (E ≥ 400, with eep ≥ 99%) compared with free r27RCL (E = 3.7, with eep = 56%) under non-aqueous conditions. Lipase r27RCL immobilized into mesoporous silica materials modified with octadecyl could be reused for at least 5 cycles without significant loss of activity and enantioselectivity. Our research suggests that lipase r27RCL immobilized into mesoporous silica materials modified with octadecyl is an attractive biocatalyst for catalysis under non-aqueous conditions.


 Please wait while we load your content...
Please wait while we load your content...