Superaerophilic copper nanowires for efficient and switchable CO2 electroreduction†
Abstract
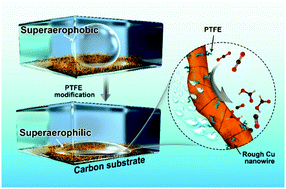
Copper is one of the most efficient electrocatalysts for switchable carbon dioxide conversion, but the design of an advanced Cu-based catalyst with high selectivity while suppressing hydrogen evolution remains a great challenge. Herein, we use Cu nanowires (Cu NWs) as the starting materials and polytetrafluoroethylene (PTFE) as the surface modifier to make a superaerophilic electrode using a wettability control strategy. This strategy allows tuning of the selectivity of the CO2 reduction reaction (CO2RR) and a decrease of the hydrogen evolution rate simultaneously by facilitating the supply of CO2 reactants and inhibiting the adsorption of water (protons). The transferring point from a pinning to bursting state turned out to be the optimized condition leading to the highest CO2RR faradaic efficiency without significant interference of current density. The optimized superaerophilic Cu NW catalyst showed CO-selectivity with a Faraday efficiency of 71% at −0.4 V vs. RHE and HCOOH-selectivity with a Faraday efficiency of 68% at −0.6 V vs. RHE. Moreover, the accelerated gas and ion diffusion and homogenized reactions also avoided accumulative damage on the surface of the Cu NWs and enhanced the stability of the Cu catalyst. This wettability tuning strategy provides a facile and efficient way to optimize the gas and ion diffusion layers, therefore promoting the performance of the CO2RR. This strategy potentially can be extended to the design of other gas consumption electrocatalysts.
- This article is part of the themed collection: Research selection from the NCNST


 Please wait while we load your content...
Please wait while we load your content...