Clioquinol induces S-phase cell cycle arrest through the elevation of the calcium level in human neurotypic SH-SY5Y cells
Abstract
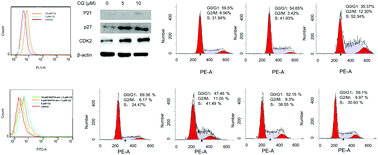
Clioquinol is recently considered to be the most promising drug for treating cancer and neurodegenerative diseases. However, its mode of action varies from different disease models. In this study, we found that clioquinol inhibited cell growth in human neurotypic SHSY-5Y cells, which was attributed to both S-phase cell-cycle arrest and autophagic cell death. Clioquinol increased the intracellular contents of iron and zinc as well as calcium as measured by ICP-AES. Staining of Fluo-3 confirmed an increase in the level of calcium. Analysis of the metal-binding ability of clioquinol showed that it was not a chelating agent of calcium ions and the elevation of intracellular calcium content is not achieved by clioquinol as an ionophore. CaCl2 could simulate or even aggravate the cytotoxicity of clioquinol and it increased S-phase cell cycle arrest induced by clioquinol in a concentration dependent manner. Staining of acridine orange demonstrated that autophagy induced by clioquinol was not affected by addition of calcium ions. In contrast, the intracellular calcium ion chelator BAPTA-am abolished the clioquinol-induced S phase arrest and reduced the cell death caused by clioquinol. The WB assay of cell cycle-related proteins (CDK2, p21 and p27) further confirmed that S phase arrest is positively correlated with intracellular calcium elevation, which was due to the alterations of the mRNA and protein levels of calcium pumps (SERCA and SPCA). Taken together, these data indicate that clioquinol regulates the level of intracellular calcium ions to induce S-phase cell cycle arrest in human SH-SY5Y cells. Our results demonstrate for the first time that an increase of intracellular calcium content is one of the mechanisms of clioquinol in the inhibition of human neurotypic SHSY-5Y cells.


 Please wait while we load your content...
Please wait while we load your content...