Cobalt(ii) cation binding by proteins†
Abstract
Herein, a set of non-homologous proteins (238) that could bind the cobalt(II) cations was selected from all the available Protein Data Bank structures with Co2+ cations. The secondary structure motifs around the amino acid residues that most frequently bind the Co2+ cations (His, Asp, and Glu) as well as the amino acid contents of the inner and outer spheres of complexes were studied. The residues forming coordination bonds to Co2+ (from the inner spheres of the complexes) are overrepresented in the regions of random coil between two β strands, between a β strand and α helix, and in all types of β strands, except that situated between an α helix and β strand. The residues situated at a distance of less than 5 Å from the Co2+ cations, but unable to form coordination bond to them (from the outer spheres of the complexes), are overrepresented in the regions of coil between the β strand and α helix and between two β strands. The data obtained for the Co2+ binding sites was compared with the data obtained for the Mg2+ and Mn2+ binding sites. Although the preferable motifs of the secondary structure for Co2+ binding (beta strand–loop–beta strand and beta strand–loop–alpha helix) are the same as those for Mg2+ and Mn2+, there are some differences in the amino acid contents of the inner and outer spheres of these complexes. The Co2+ cations are preferably coordinated by a combination of His and Glu residues, whereas the Mn2+ and Mg2+ cations prefer a combination of His and Asp and just Asp residues, respectively. As a result, two computer algorithms were developed that could evaluate the possibility of Mg2+ and Mn2+ replacement by the Co2+ cations (chemres.bsmu.by). These algorithms should help to investigate the pathogenesis of cobalt intoxication occurring in patients with cobalt-containing artificial joints.
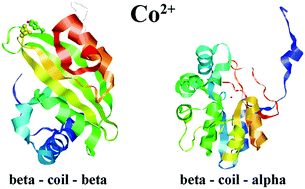


 Please wait while we load your content...
Please wait while we load your content...