Structure and anticancer activities of four Cu(ii) complexes bearing tropolone†
Abstract
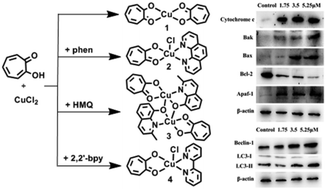
Agents inducing apoptosis and autophagic death could be effective chemotherapeutic drugs. In this work, four novel Cu(II) complexes formulated as [CuL2] (1), [Cu(phen)LCl]·0.5H2O (2), [Cu2(MQ)2L2] (3) and [Cu(2,2′-bpy)LCl]·H2O (4) (phen = 1,10-Phenanthroline, HMQ = 8-hydroxy-2-methylquinoline, 2,2′-bpy = 2,2′-bipyridine) were prepared from the reactions of copper(II) chloride with tropolone (HL) in the absence or presence of different ancillary ligands. The solution state structures of 1, 2 and 4 agree well with their solid state structures. Complex 3 presents a dimer structure in the solid state, however, a monomer structure in the solution state. It was shown that all of these complexes are stable under experimental conditions and bind to DNA in an intercalative mode with the binding constant Kb values of 1.05 × 103, 2.57 × 103, 2.53 × 103 and 2.26 × 103 L mol−1 for complexes 1, 2, 3 and 4, respectively. The anti-proliferative tests against cultured human cancer cell lines (A549, Bel-7402, MGC80-3, T24, SK-OV-3, and NCI-H460) in vitro revealed cytotoxic activities for these complexes, which are much better than those for all ligands in these complexes, as well as that for cis-platin. After a careful comparison, the cytotoxic activity of complex 2 against MGC80-3 cells in vitro (IC50 = 3.5 ± 0.9 μM for 2 and 18.0 ± 1.2 for cis-platin) was further investigated in detail as an example. 2 induces the apoptosis of MGC80-3 through a caspase-dependent mitochondrion pathway and can also induce autophagy, which revealed a certain anticancer activity for complex 2.


 Please wait while we load your content...
Please wait while we load your content...