Low-molecular-mass iron complexes in blood plasma of iron-deficient pigs do not originate directly from nutrient iron†
Abstract
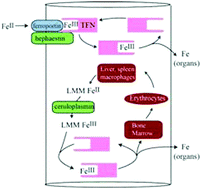
Nutrient iron entering the blood binds transferrin (TFN)d, which delivers iron to cells in the body. In healthy individuals, ∼30% of TFN is iron-bound while the remainder is unbound (apo-TFN). TFN saturates the plasma of individuals with iron-overload diseases such as hereditary hemochromatosis, prompting release of a poorly-defined low-molecular-mass (LMM) iron species called non-transferrin-bound iron (NTBI). An experiment was devised to directly detect NTBI in plasma of iron-deficient pigs and to assess the role of the liver which is known to bind NTBI. Catheters were surgically installed in the portal vein (PV) and either the caudal vena cava or the cranial vena cava. After the animals recovered, 57Fe II ascorbate was injected into the stomach via a feeding tube. Blood was removed through the catheters before and after injection; plasma became 57Fe-enriched after injection. 57Fe-enriched plasma was passed through a 10 kDa cutoff membrane and the flow-through solution (FTS) was subjected to size-exclusion liquid chromatography (LC). The eluent flowed into an ICP-MS where 56Fe and 57Fe were detected. Low-intensity iron peaks with masses of 400–1600 Da were observed, but none became enriched in 57Fe after injection. Rather, the injected 57Fe bound to apo-TFN. Viewed naively, this implies that nutrient-derived 57Fe in healthy mammals passes from the intestines to apo-TFN without first entering the blood as a LMM intermediate. In this case, nutrient iron exported from intestinal enterocytes of healthy individuals may quickly bind apo-TFN such that LMM iron species do not accumulate in blood plasma. Some 57Fe from the FTS may have adsorbed onto the column. In any event, the LMM iron species in plasma that eluted from the column must have originated from iron stored within the body, perhaps in macrophages – not directly from nutrient iron absorption. The liver absorbed and released LMM iron species, but the effect was modest, consistent with its role as a dynamic iron buffer. Passage through the liver also altered the distribution of different forms of TFN present in the PV.


 Please wait while we load your content...
Please wait while we load your content...