Characterization and identification of selected metal-binding biomolecules from hepatic and gill cytosols of Vardar chub (Squalius vardarensis Karaman, 1928) using various techniques of liquid chromatography and mass spectrometry†
Abstract
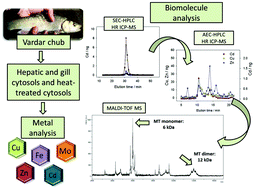
Metals play crucial physiological roles, but they can also cause irreparable toxic effects through binding to important cellular biomolecules in aquatic organisms. The aim of this study was to determine the exact molecular masses and to identify several selected metal-binding biomolecules in hepatic and gill cytosols of Vardar chub (Squalius vardarensis Karaman, 1928). Methods applied for the achievement of this goal were SEC-AEC-HPLC for two-dimensional separation of cytosolic biomolecules, HR ICP-MS for metal measurements, and mass spectrometry (MALDI-TOF-MS and LC-MS/MS) for biomolecule mass determination and identification. The analyzed biomolecules included: Fe-binding biomolecules, which were identified as hemoglobin subunit β in the liver (molecular masses of ∼15 kDa), and hemoglobin subunits α and β in the gills (molecular masses of ∼11 kDa, ∼13 kDa and ∼15 kDa); heat-stable Cd-binding biomolecules, which were identified as MT isoforms MT-I and MT-II (molecular mass of ∼6.0 kDa in both liver and gills, and an additional 4.9 kDa isoform in the gills); and heat-stable Mo-binding biomolecules of molecular masses equal to 3.3 kDa (in the gills) and 8.5 kDa (in the liver). An important finding of this study was the obvious presence of different isoforms of the same biomolecules in the liver and gills. This was, among others, manifested through the absence of Zn binding to MTs only in the gills, indicating that the same type of biomolecule can be responsible for different functions in different organs. Thus, for better understanding of metal behaviour in aquatic organisms, it is crucial to identify cellular metal-binding biomolecules and their functions.


 Please wait while we load your content...
Please wait while we load your content...