Active uptake of hydrophilic copper complex Cu(ii)–TETA in primary cultures of neonatal rat cardiomyocytes†
Abstract
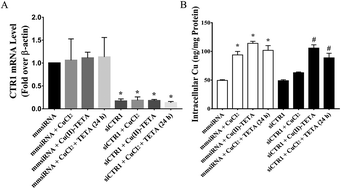
Myocardial ischemia leads to copper efflux from the heart. The ischemic tissue with a low copper content fails to take up copper from the circulation even under the conditions of serum copper elevation. Cardiac copper repletion thus requires other available forms of this element than those currently known to bind to copper transport proteins. The copper complex of triethylenetetramine (TETA) is a metabolite of TETA, which has the potential to increase cardiac copper content in vivo. In the present study, we synthesized Cu(II)–TETA, analyzed its crystal structure, and demonstrated the role of this compound in facilitating copper accumulation in primary cultures of neonatal rat cardiomyocytes. The Cu(II)–TETA compound formed a square pyramidal chloride salt [Cu(TETA)Cl]Cl structure, which dissociates from chloride in aqueous solution to yield the four-coordinate dication Cu(II)–TETA. Cu(II)–TETA was accumulated as an intact compound in cardiomyocytes. Analysis from time-dependent copper accumulation in cardiomyocytes defined a different dynamic process in copper uptake between Cu(II)–TETA and CuCl2 exposure. An additive copper accumulation in cardiomyocytes was found when the cells were exposed to both CuCl2 and Cu(II)–TETA. Gene silencing of copper transport 1 (CTR1) did not affect cross-membrane transportation of Cu(II)–TETA, but inhibited copper cellular accumulation from CuCl2. Furthermore, the uptake of Cu(II)–TETA by cardiomyocytes was ATP-dependent. It is thus concluded that the formation of Cu(II)–TETA facilitates copper accumulation in cardiomyocytes through an active CTR1-independent transportation process.
- This article is part of the themed collection: Metallomics Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...