Metabolic response to Parkinson's disease recapitulated by the haploinsufficient diploid yeast cells hemizygous for the adrenodoxin reductase gene†
Abstract
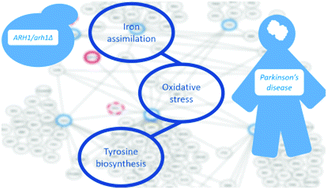
Adrenodoxin reductase, a widely conserved mitochondrial P450 protein, catalyses essential steps in steroid hormone biosynthesis and is highly expressed in the adrenal cortex. The yeast adrenodoxin reductase homolog, Arh1p, is involved in cytoplasmic and mitochondrial iron homeostasis and is required for activity of enzymes containing an Fe–S cluster. In this paper, we investigated the response of yeast to the loss of a single copy of ARH1, an oxidoreductase of the mitochondrial inner membrane, which is among the few mitochondrial proteins that is essential for viability in yeast. The phenotypic, transcriptional, proteomic, and metabolic landscape indicated that Saccharomyces cerevisiae successfully adapted to this loss, displaying an apparently dosage-insensitive cellular response. However, a considered investigation of transcriptional regulation in ARH1-impaired yeast highlighted that a significant hierarchical reorganisation occurred, involving the iron assimilation and tyrosine biosynthetic processes. The interconnected roles of the iron and tyrosine pathways, coupled with oxidative processes, are of interest beyond yeast since they are involved in dopaminergic neurodegeneration associated with Parkinson's disease. The identification of similar responses in yeast, albeit preliminary, suggests that this simple eukaryote could have potential as a model system for investigating the regulatory mechanisms leading to the initiation and progression of early disease responses in humans.


 Please wait while we load your content...
Please wait while we load your content...