Is protein context responsible for peptide-mediated interactions?†
Abstract
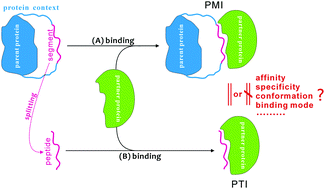
Many cell signaling pathways are orchestrated by the weak, transient, and reversible protein–protein interactions that are mediated by the binding of a short peptide segment in one protein (parent protein) to a globular domain in another (partner protein), known as peptide-mediated interactions (PMIs). Previous studies normally had an implicit hypothesis that a PMI is functionally equivalent or analogous to the protein–peptide interaction (PTI) involved in the PMI system, while ignoring parent context contribution to the peptide binding. Here, we perform a systematic investigation on the reasonability and applicability of the hypothesis at structural, energetic and dynamic levels. It is revealed that the context impacts PMIs primarily through conformational constraint of the peptide segments, which can (i) reduce the peptide flexibility and disorder in an unbound state, (ii) help the peptide conformational selection to fit the active pocket of partner proteins, and (iii) enhance the peptide packing tightness against the partners. Long, unstructured and/or middle-located peptide segments seem to be more vulnerable to their context than short, structured and/or terminal ones. The context is found to moderately or considerably improve both the binding affinity and specificity of PMIs as compared to their PTI counterparts; with the context support a peptide segment can contribute to ∼30–60% total binding energy of the whole PMI system, whereas the contribution is reduced to ∼5–50% when the context constraint is released. In addition, we also observe that peptide selectivity is largely impaired or even reversed upon stripping of their parent context (global selectivity decreases from 34.2 to 1.7-fold), by examining the crystal structures of full-length Src family kinases in an autoinhibitory state. Instead of the direct interaction and desolvation that are primarily concerned in traditional studies, peptide flexibility and the entropy penalty should also play a crucial role in the context effect on PMIs. Overall, we suggest that the context factor should not be ignored in most cases, particularly those with peptide segments that are long, highly disordered, and/or located at the middle region of their parent proteins.


 Please wait while we load your content...
Please wait while we load your content...